Report: Handmade Emulsion with Sight Unseen
Below is a brief report of our Handmade Emulsion workshop held in Baltimore, Maryland on July 12th & 13th at Current Space and in collaboration with Sight Unseen. The workshop was lead by Process Reversal members Andy Busti and Kevin Rice.
In this workshop, a small fraction of the theories and practices relating to emulsion making were discussed and implemented in the generation of two different emulsions. These emulsions were then coated on to 16mm clear acetate leader and used for in camera photography. Below are the notes relating to each of these emulsions and their resulting images.
Emulsion 1
Below is the formula for our first emulsion, E1, a neutral bromide emulsion. The materials for the formula were sourced from various suppliers and locations: the potassium bromide came from the Photographers Formulary in Montana, the silver nitrate from ArtCraft Chemicals in New York, the distilled water from a local 7 Eleven, and finally the Knox brand food gelatin from a grocery store in Boulder, CO.
| TECHNICAL DATA | E1 |
| Type | Single Jet, Bromide, Neutral |
| Approx. Shelf Life | 3.00 months |
| Coating Capacity | +/- 25.00 ft² |
| pH | — |
| SOLUTION A | |
| Distilled Water | 63.0 ml |
| Potassium Bromide [KBr] | 8.0 g |
| Active Gelatin | 10.0g |
| SOLUTION B | |
| Distilled Water | 63.0 ml |
| Silver Nitrate [AgNO3] | 10.0 g |
Table 1 – E1 Technical Specifications and Formula
The formulas constituents were divided into two solutions: Solution A contained the gelatin and bromide salt whereas solution B contained the silver salt. Both solutions were mixed separately from each and with strict care so as to avoid any cross contamination. This process was carried out at room temperature so as to avoid excessive condensation of the water from the solutions, after which they were brought to emulsification temperature (approx. 60 C). Solution A was contained within a sterilized 250.0 ml pyrex beaker whose temperature was maintained by a dual hot plate / magnetic stirrer. Solution B was also contained within a sterilized, 100.0 ml, pyrex beaker but was heated in a water bath before being divided between two plastic syringes fitted with very fine nozzles. At this point, the room lights were taken out and the emulsification process commenced under safelight.
Emulsification
Emulsification was carried out as a sub-surface, single jet addition across two stages: the first stage saw the rapid addition of half of the silver nitrate into the halogen rich gelatin in an attempt to create an environment optimized for critical nucleation of the silver halide crystals. During this first addition, a malfunction occurred with the syringe tip which became dislodged from the syringe and fell into the solution. This was ignored momentarily and the emulsion was allowed to ripen for 15 minutes, without agitation, before the tip was removed using a sterilized glass stirring rod. Following this, the next stage of emulsification commenced whereby the second half of the silver nitrate solution was slowly added across 20 minutes in an attempt to produce fine precipitates that would be more readily nucleated by the larger crystals precipitated through the first stage. At the conclusion of this second addition, the emulsion was allowed to sit for approx. 5 minutes without agitation before being chilled rapidly.

Image 4 – One of the syringes and nozzles used to create the jet for solution B. The syringe was sourced from a gardening supply store whereas the nozzle was purchased seperately from an art supply store, both in Denver, CO.
Washing
In order to remove the by-products produced in the emulsification stage (namely for coating reasons), the emulsion was washed in the old tradition of “noodling.” This was done by pressing the emulsion through a fiberglass mesh and catching the fine strands in 4 layers of unbleached cheesecloth fixed to the mouth of a bucket. Ice water was then repeatedly poured over the noodles, using the fiberglass mesh to filter out any ice cubes, until about 1.5 gallons of water had been poured across the emulsion. After this, 0.5 gallons of cold distilled water was used to complete the washing of the emulsion before being carefully consolidated into the original emulsification vessel and remelted.
Chemical & Spectral Sensitization
The emulsion was not treated with any sensitizers other than those initially present in the gelatin.
Finals & Coating
After remelting the emulsion, 20.0 ml of Kodak photo-flo 2000 was added as a surfactant to aid in the coating process. This was the only final addition to the emulsion.
Coating was performed under a safe light using a film rewind to feed the pre-subbed, 16mm acetate film base onto a coating wheel. Participants worked together to maintain tension across the system and apply the emulsion using two different styles of brushes: (1) A ¾” Hake style brush and (2) A ¾” Blick Studio synthetic brush, the latter of which was found to produce the “best” coating results.

Image 5 – A Hake brush, the preferred method of coating. The brushes used during the workshop were purchased at a local art supply store.
After the first coat was complete, the film was allowed to dry for over an hour before a second coat was applied using an airbrush. Finally, the film was allowed to sit in darkness overnight to dry to completion.
Photography & Development
The following day, the coated film was wound onto a daylight spool for in-camera photography with a Bolex Rex 5. Half of the participants went out to photograph local scenes with the emulsion while the second half began producing the second emulsion discussed below. Exposure for the emulsion was determined using a spot meter with the film rated at 10 (purely out of guesstimation) and only 50 feet was photographed to begin with.
| TECHNICAL DATA | KODAK DEKTOL |
| Type | Developer (MQ based, High Energy) |
| Approx. Shelf Life | 3.00 months |
| Capacity | +/- 30.00 ft² per. 1,000.00 ml |
| pH | 11.0 |
| STOCK SOLUTION | |
| Water @ 52.0 C | 750.0 ml |
| Metol [C7H9NO · 1/2H2SO4] | 3.0 g |
| Sodium Sulfite, Anhydrous [Na2SO3] | 45.0 g |
| Hydroquinone [C6H4(OH)2] | 12.0 g |
| Sodium Carbonate (Monohydrate) [Na2CO3· 1H20] | 80.0 g |
| Potassium Bromide [KBr] | 2.0 g |
| Water to make… | 1,000.0 ml |
Table 2 – Approx. technical specifications and formula for Kodak Dektol
Development of the emulsion was carried out in a 50 foot LOMO tank using Kodak Dektol, a high energy print developer, with a development time of 2 minutes. To avoid damaging the film, agitation was not used. Following development, the film was then rinse delicately and fixed in a hardening fixer for approx. 2 minutes before a brief final rinse. This first run was then hung to dry while the second half of the film was photographed.
Analysis
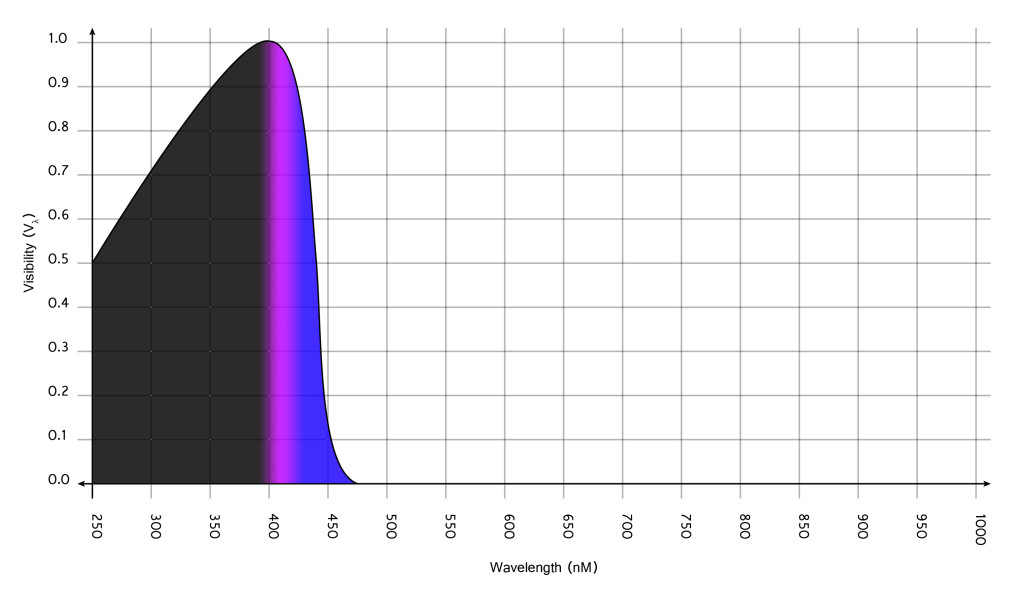
While equipment was not available to us for adequately test the sensitometric properties of the emulsion, we can make a few assumptions: First, we know that no spectral sensitizers were used and thus the emulsions sensitivity can be defined as class 1, sensitive only to the native absorption properties of the silver bromide crystals. Accordingly, the emulsion was primarily sensitize to the UV spectrum as well as some portions of violet and blue light.
When exposed under daylight, the emulsion exhibited a sensitivity equivalent to International Standards Organization (ISO) 10, and is therefore considered a very slow, and thus fine fine grained, emulsion. Additionally, based on the visual appearance of the negative, the emulsion appears to have been able to render scenes with a 5-7 stop range, even when processed with a high energy developer (i.e. Kodak Dektol), and thus can be classified as a “low contrast” emulsion. This, coupled with the knowledge of how the film was emulsified, indicates that the emulsion grains were polydispersed and thus exhibited a great deal of variation in size. Finally, given the halogen and the emulsification methodology, we can make the assumption that the crystal habit yielded a fairly decent portion of tabular grains, along with various other grain structures, most of which probably did not exhibit any properties of photosensitivity (i.e. a low efficiency emulsion). Sensitivity was also certainly altered by the choice of gelatin, which in this case was not inert. In that sense, it can be loosely categorized as an “active” gelatin, and thus would have contributed some portion of sulfides and other constituents that would have increased the sensitivity.
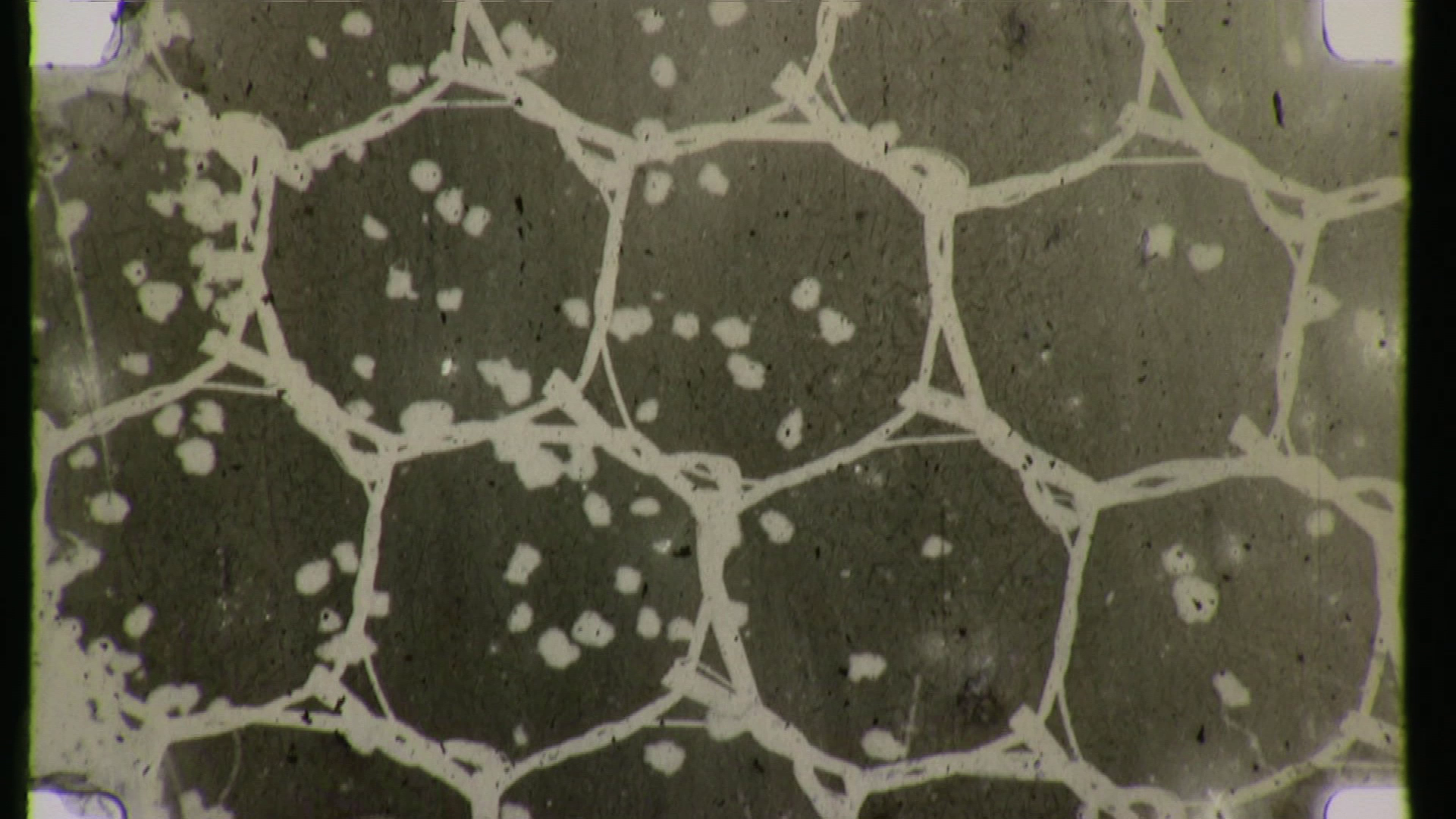
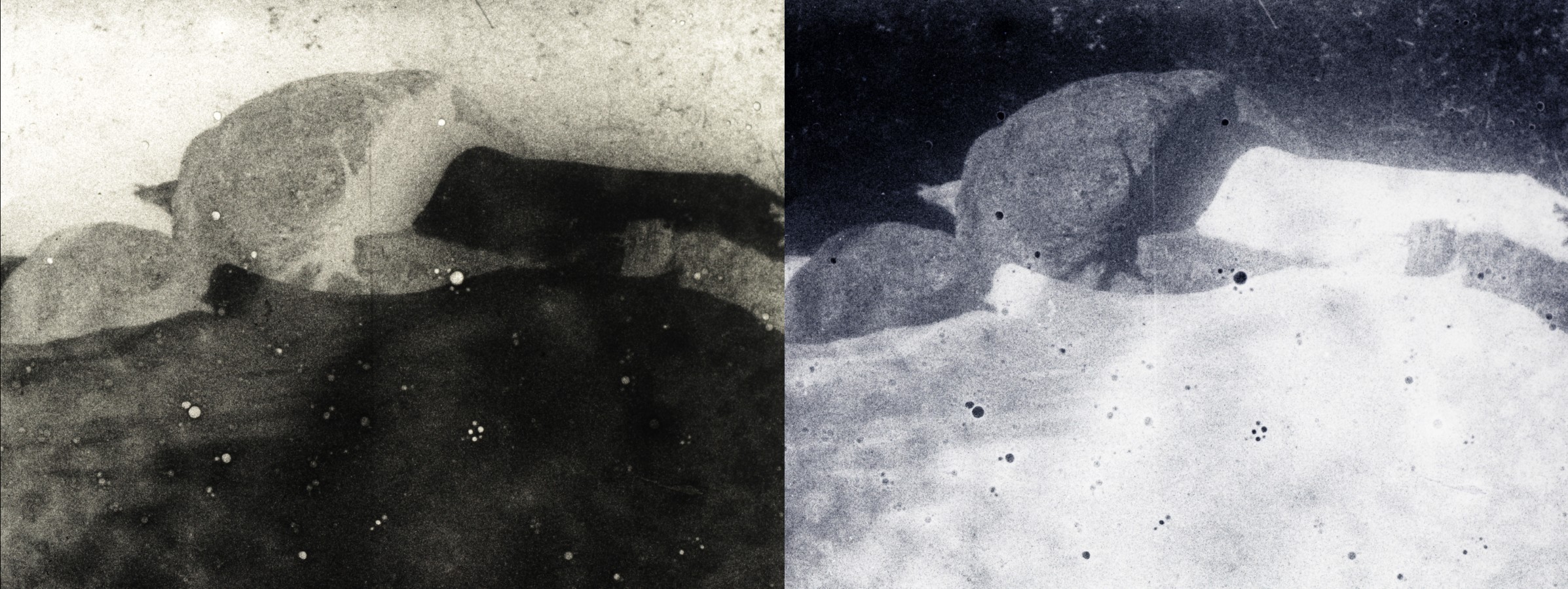
Below is documentation of the camera original negative, as well as a digitally inverted timed “print” of the footage. Additionally, some high resolution blow ups have been chosen for the gallery below, including high magnifications of the granularity of the film, which can be characterized as very high, most likely because of grain migration and clumping during development and not because of the grain structure…
Video 1 – The camera original negative; Due to the host sites video compression, it is suggested that the original file be downloaded for optimal viewing.
Gallery 1 – Camera original negative; select frames. A ZIP file with higher resolution files can be downloaded here.
Contact Prints
Two contact prints were also struck from the original negative by workshop participants. This was done by bi-packing a daylight spool with both the original negative and our raw print stock, and then exposing it without a lens and with an enlarger as a light source. Two different print stocks were used: Kodak 7302 Fine Grain positive Release and Kodak 7363 High Contrast Positive II.
The 7363 print was mostly over exposed and highlighted many of the emulsion coating defects, making the imagery extremely “abstract.” The 7302 print, on the other hand, was very well exposed and resolved many of the details of both the images and the emulsion coating. In both cases, only the first 50 feet of the camera original was printed…
Video 2 – The bolex contact prints struck during the workshop. Again, due to the host sites video compression, it is suggested that the original file be downloaded for optimal viewing.
Emulsion 2
Below is the formula for the second emulsion formula, E2, a neutral chloride emulsion. The recipe utilized much of the same constituents as the first emulsion, except that sodium chloride was used in place of potassium bromide as a source for the halogen. This was sourced from The Science Company in Denver, CO.
| TECHNICAL DATA | E2 |
| Type | Single Jet, Chloride, Neutral |
| Approx. Shelf Life | 3.00 months |
| Coating Capacity | +/- 75.00 ft² |
| pH | — |
| SOLUTION A | |
| Distilled Water | 500.00 ml |
| Sodium Chloride [NaCl] | 12.0 g |
| Active Gelatin | 90.0g |
| SOLUTION B | |
| Distilled Water | 100.0 ml |
| Silver Nitrate [AgNO3] | 25.0 g |
Table 3 – E2 technical specifications and formula
Emulsification
Emulsification was carried out nearly identically to that of the first emulsion, except that participants used a stainless steel film processing tank to act as the emulsification vessel. During each addition, one participant would stir the emulsion with a glass rod while another injected the silver nitrate solution.
Washing
This emulsion was not washed in order to save time. This should not have had an appreciable effect on the sensitivity of the emulsion, but may have resulted in problems with the coating as the salt by-products precipitated out of the emulsion.
Chemical and Spectral Sensitization
Again, no chemical or spectral sensitizers were added to the emulsion.
Finals & Coating
Approx. 30.0 ml of Kodak Photo-Flo 2000 was added as a surfactant to aid in the coating process. This was the only final addition to the emulsion. The same coating arrangement was used as with the first emulsion, except that both coats were performed using hake brushes only. Additionally, participants coated several sheets of paper for use in photographic enlargements.
Photography & Development
By the time the film was dry, there was no longer sufficient daylight to photograph the roll; this was instead given up to the workshop participants, and as of this writing, the roll has not been transferred. However, some of the coated sheets were used to create enlargements which were then processed in the same manner as the first emulsion. No scans of these enlargements has been performed as of this writing…
Analysis
Because there wasn’t enough time to really adequately photograph and develop the emulsion, analysis is limited. Based on the sheets that were processed, the emulsion appeared to be very, very slow (equivalent to 1 ISO) However, this could be attributed to a number of factors; for one, chloride emulsion are almost exclusively sensitive to the UV spectrum. Since the exposure source for the enlargements was primarily tungsten, the amount of light absorbed by the emulsion would have been limited to a fraction of this light, thus resulting in very long exposure times. Second, the sheets were only coated once which did not appear to be able to yield significant density even with extensive exposure and development times. Thus a second coat was applied to the sheets, but there was not enough time to expose these and gauge the difference.
Extended Resources:
A PDF of some basic resources, including conversions and formulas, can be downloaded here. In addition to this, the following resources can also be explored…
Literature
The following selection of literature is a fairly comprehensive list of text relating to the process of emulsion making and it’s related sciences. Some of these text are highly recommended and have been indicated as such with an asterisk (*) Additionally, any hyperlinked text are available to download from Process Reversals website at http://processreversal.org/literary-resources/
Practical Guides to Emulsion Making
These text are recommended for those interested in practical examples and insights into creating emulsions…
- Re:inventing the Pioneers: Film Experiments on Handmade Silver Gelatin Emulsion (Urulus, Esther – 2013)*
- Making, Coating and Processing a Simple Gelatin Emulsion (Osterman, Mark – 2007)
- Photographic Emulsion Making, Coating & Testing (Mowery, Ron – 2009)
- Dye Transfer Materials (Browning, James – 1998)
- Silver Gelatin: A User’s Guide to Liquid Photographic Emulsions (Reed, Martin; Jones, Sarah – 2001)
Historic Emulsion Text
These text are recommended for those seeking historical context to the emulsion making process. Many of them will discuss emulsion making techniques and formulas, but it’s important to understand that any literature dealing with emulsion published before the 1940’s assumes the use of photograde ACTIVE gelatin, which is no longer produced. Everything following that assumes the use of inert gelatin.
- The Silver Sunbeam (Towler, John – 1864)
- Modern Dry Plates (Eder, J.M. – 1881)
- Photography with Emulsions (Abney, William De W. – 1885)
- Dry Plate Making for Amateurs (Sinclair, George L. – 1886)
- Photographic Emulsions (Wall, E.J. – 1929)
- The Photographic Emulsion (Carroll, B.H.; Hubbard, D.; Kretschman, C.M. -1934)
- Photographic Emulsion Technique (Baker, T. Thorne – 1941)*
- The Theory of the Photographic Process (Mees, C.E. Kenneth – 1942)
- Making Kodak Film (Shanebrook, Robert L. – 2012)
Emulsion Chemistry & Theory
These text are recommended for those interested in studying the theory and science of emulsion making.
- Film Coating Theory (Deryagin, B.V.; Levi, S.M. – 1964)
- Making and Coating Photographic Emulsions (Zelikman, V. L.; Levi, S. M. – 1964)
- Photographic Gelatin (Croome, R.J.; Clegg, F.G. – 1965)
- Photographic Emulsion Chemistry (Duffin, G.F. – 1966)*
- Photographic Sensitivity: Theory and Mechanisms (Tadaaki, Tani – 1995)*
Sensitometry
- General Sensitometry (Gorokhovskii, Yu. N.; Levenberg, T.M. – 1965)
- Photographic Sensitivity: Theory and Mechanisms (Tadaaki, Tani – 1995)
Photochemistry
- Photographic Processing Chemistry (Mason, L.F.A. – 1966)*
- Motion Picture and Television Film: Image Control and Processing Techniques (Corbett, D.J. – 1968)*
- Developing: The Negative Technique (Jacobson, C.I.; Jacobson, R.E. -1976)
- The Film Developing Cookbook (Tropp, Bill; Anchell, Stephen G.-1998)*
Reference Books
These books are helpful as references for various formulas and darkroom techniques
- Photographic Lab Handbook, 5th Edition (Carroll, John S. – 1979)
- The Darkroom Cookbook, 3rd Edition (Anchell, Stephen G. – 2009)*
Artist Run Film Labs
- Kinetica: Lieux d’Experimentations Cinematographiques en Europe (Gran Lux – 2011)
Web Resources
- Analog Photographers User Group (APUG.org)
This website houses the most popular forum relating to analog photography. While not specifically relating to motion picture film, there are many conversations and researchers of interest, including Ron Mowery (http://www.apug.org/forums/members/photo-engineer/) who is particularly informative on the theory and practice of emulsion making and has contributed heavily to the APUG subsection on Silver Gelatin Based Emulsion Making (http://www.apug.org/forums/forum205/). To subscribe to the forum, follow the instructions here: http://www.apug.org/forums/register.php - The Light Farm (thelightfarm.com)
- Film Labs (filmlabs.org)
This website houses information on various artist run film labs around the world and is also the host of the “film labs forum,” a subscription based mailing list featuring discussions on all varieties of lab related subjects. To subscribe to the forum, follow the instructions posted here: https://listes.domainepublic.net/listinfo/forum - Graphic Atlas (graphicsatlas.org)
This website is dedicated to the documentation of various photographic processes and contains a very impressive collection of articles and images relating to the identification and archival handeling of various photogrpahic images. - Molecular Weight Calculator
Chemical Suppliers
- ArtCraft Chemicals (http://www.artcraftchemicals.com/) — New York based photochemical supplier with a good selection and good prices, particularly for silver nitrate.
- The Science Company (http://www.sciencecompany.com/) — Denver based chemistry and lab equipment supplier. Higher prices than most, but convenient if working in Colorado.
- Photographers Formulary (http://stores.photoformulary.com/) — Montana based photochemical supplier with a moderate selection and good prices. Also sells kits and books.
- Nymoc Products Co. (https://plus.google.com/111988851146358298635/about?hl=en) — Toronto based chemical supplier with the widest selection at the highest cost.
Photo Credit: Margaret Rorison. For more photos of the workshop, a zip file can be downloaded here.



































I’ve been studying the process of making my own B&W film (for negatives and print paper) and have never come across the term used here “Active Gelatin”. I thought that Photo Grade Gelatin of 250 Baume was the prefered gelatin. After reading here about Active Gelatin I did a little more investigation and found a discussion on APUG regarding this subject. Here’s a link to the discuss between two members, one fellow “Photo Engineer” seems to have a good point. Could someone comment or elaborate on the subject of “Active Gelatin”
Thanks so much,
Seth Martin